



Sermorelin is a highly significant compound within the Growth Hormone axis research bracket, designed to stimulate and analyze the intricate cascades of systemic metabolic regeneration and endocrine pulsing.
| Parameter | Value |
|---|---|
| Molecular Formula | C149H246N44O42S1 |
| Molecular Weight | 3357.9 g/mol |
| Assay Purity | >99.0% |
| Appearance | Lyophilized solid |
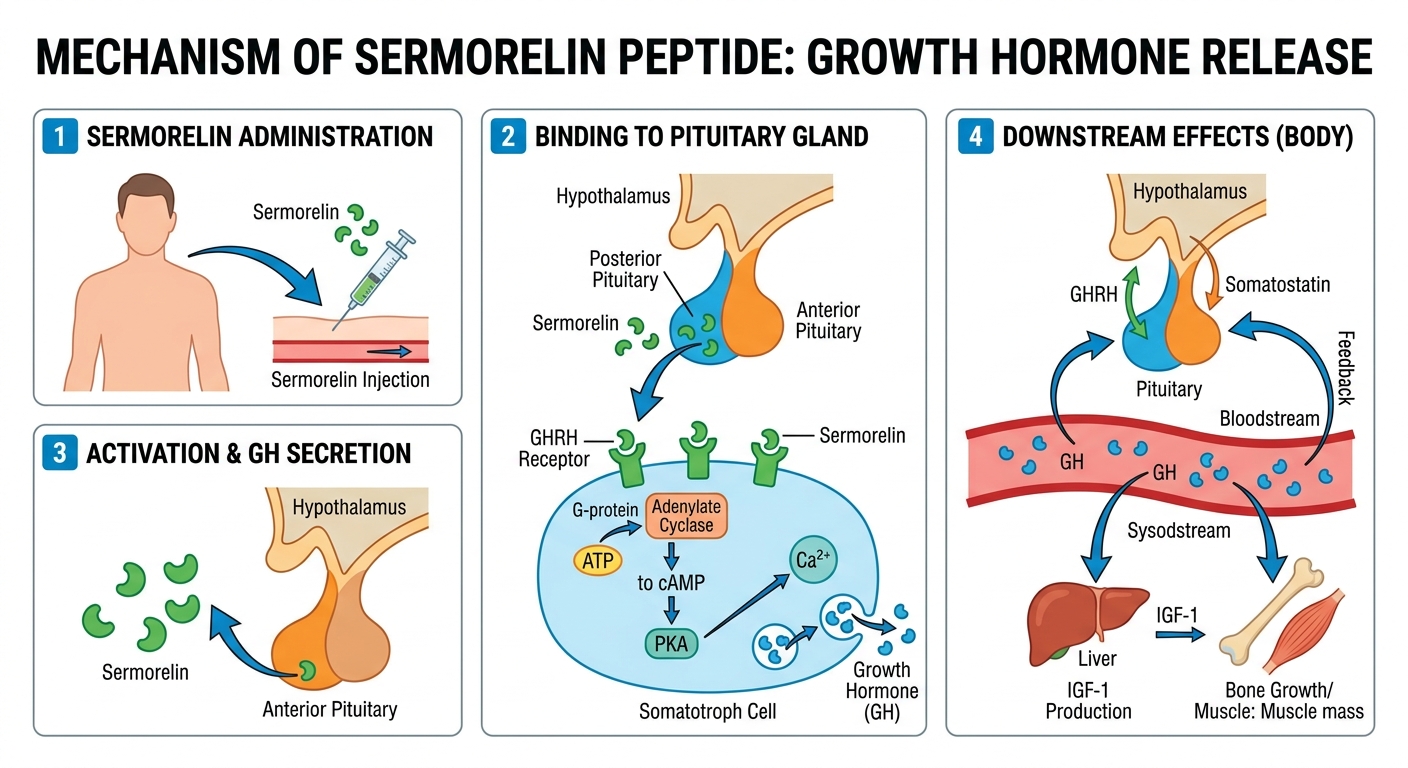
Figure 1: Conceptual Binding Mechanism of Sermorelin
The sequential action framework observed in experimental diagnostics:
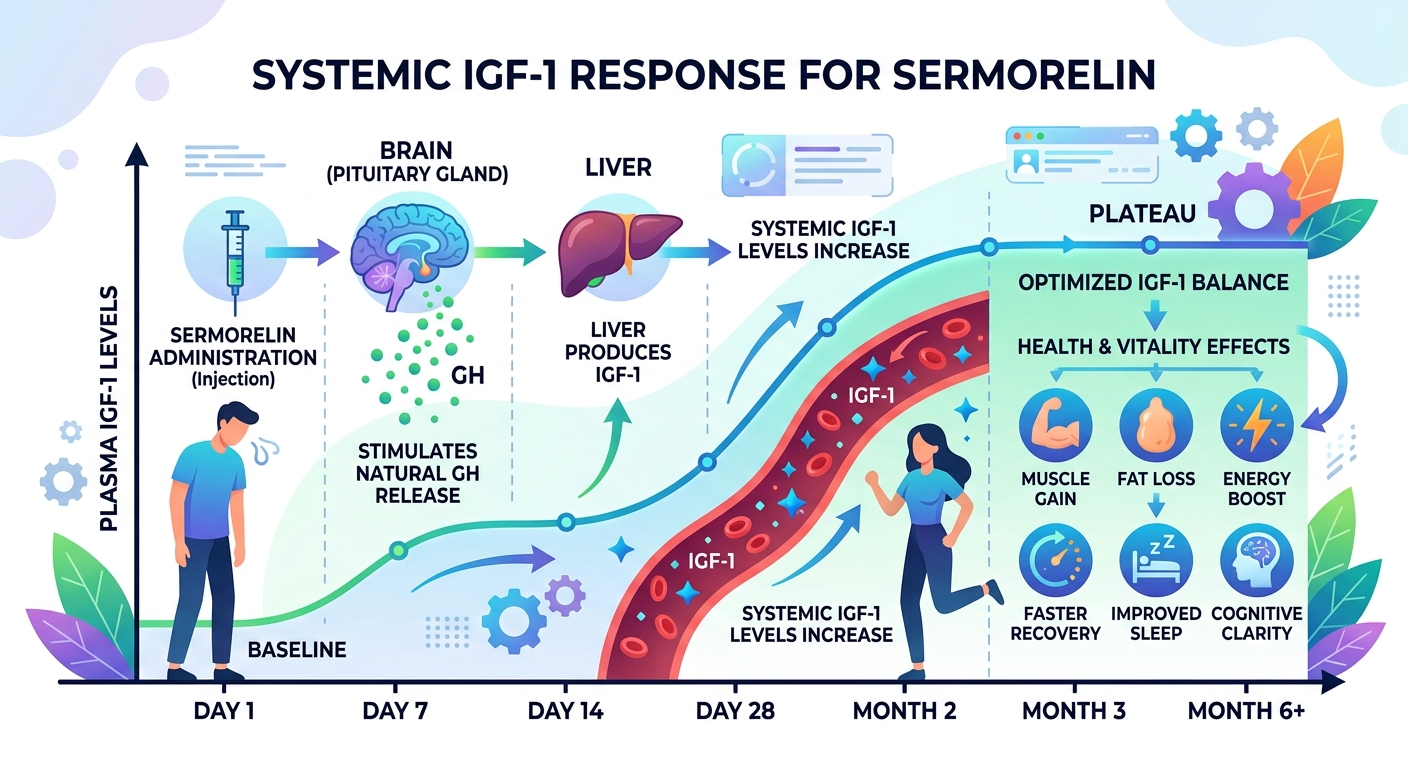
Figure 2: Empirical Systemic Response Profile
Sermorelin (Geref®) is the 29-amino acid N-terminal fragment of human growth hormone-releasing hormone (GHRH 1-44), representing the minimal sequence required for full biological activity at the GHRH receptor. The discovery that residues 1–29 retain 100% of the receptor binding and signaling potency of the full 44-amino acid peptide was a key milestone in neuroendocrine research. Sermorelin was the first GHRH analogue to receive FDA approval (1997) for diagnostic evaluation of pituitary GH secretory capacity and later for the treatment of idiopathic GH deficiency in children.
Every batch of Sermorelin is tested by an independent third-party laboratory. The Certificate of Analysis (COA) confirms identity, purity, and sterility of each lot.
COA documents are available upon request. Contact us with your order number to receive the COA for your specific batch.
High-Performance Liquid Chromatography (HPLC) is used to verify the purity of Sermorelin. Our products consistently test at 99%+ purity via reverse-phase HPLC.
HPLC chromatograms are available for each batch. Request your batch report.
Mass Spectrometry (MS) confirms the molecular weight and structural identity of Sermorelin, ensuring the compound matches its expected molecular profile.
Mass spectrometry reports are available on request. Contact support for documentation.
We've been using this for ongoing research and the results are reproducible. That's the most important thing.