


SLUP-332 Tablets represents an advanced oral delivery system, engineered to survive gastrointestinal degradation and provide systemic biological activity without the need for traditional parenteral injection.
| Parameter | Value |
|---|---|
| Molecular Formula | C28H38N4O6 |
| Molecular Mass | 526.6 g/mol |
| Assay Purity | >98.0% |
| Appearance | White round compressed tablets |
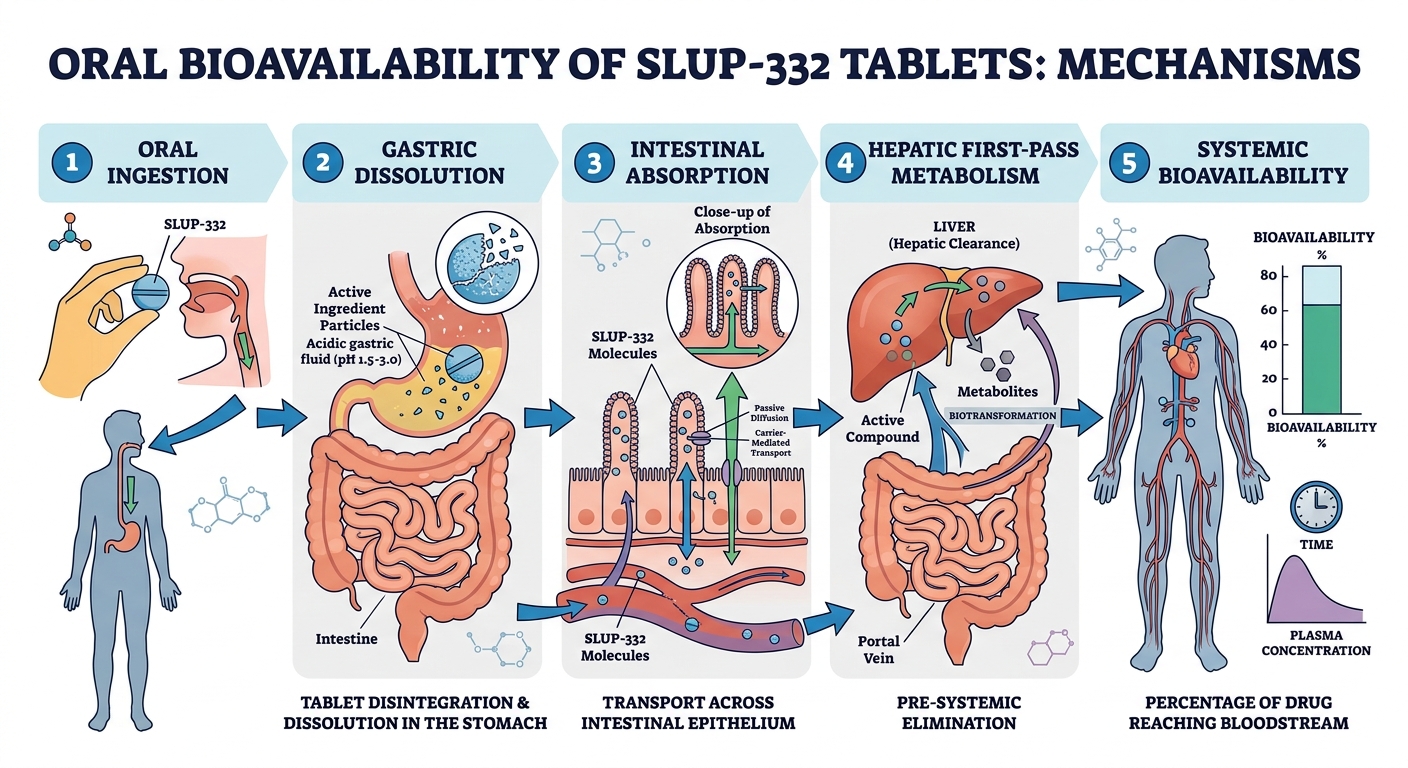
Figure 1: SLUP-332 Tablets GI Tract Dissolution & Absorption
Detailed cascade of actions from ingestion to systemic effect:
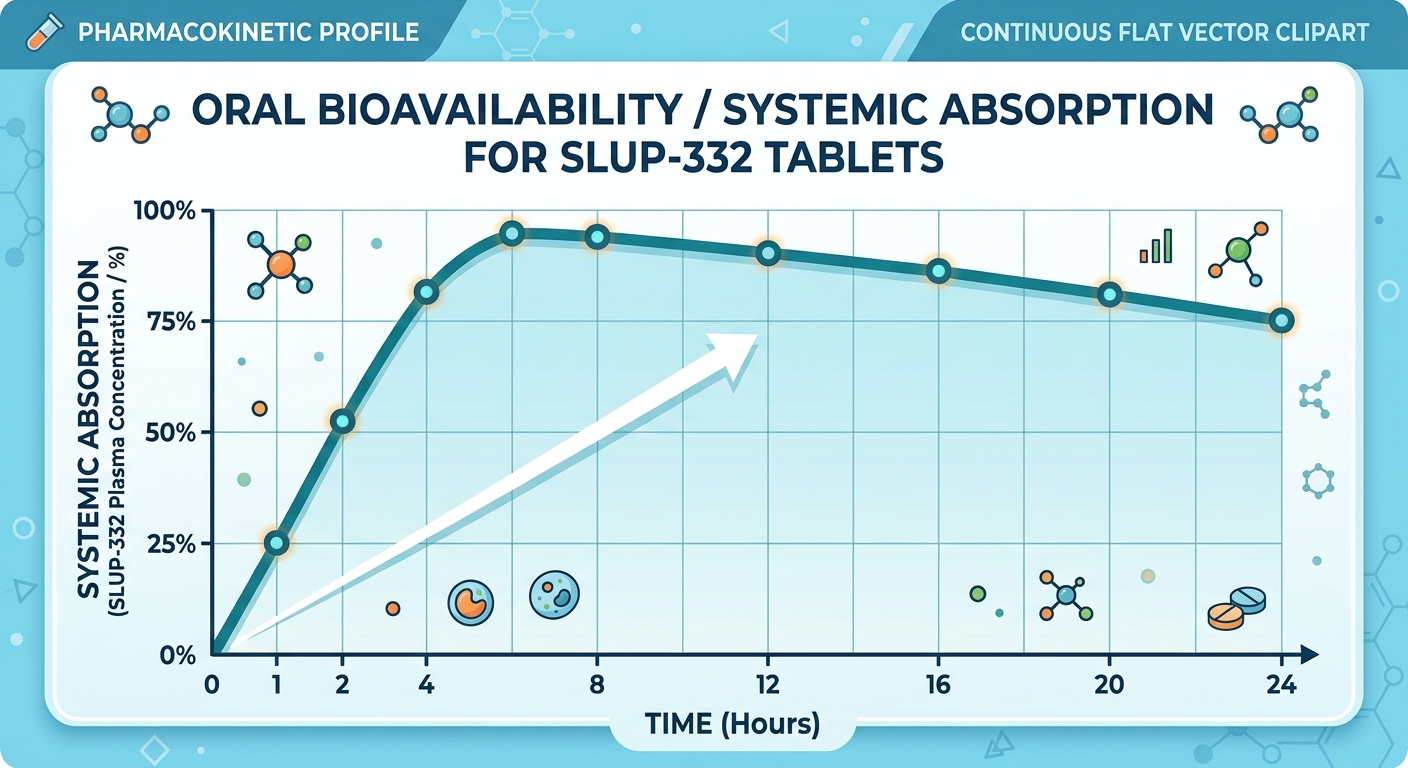
Figure 2: Rate of Oral Systemic Bioavailability
SLUP-332 is a non-steroidal selective androgen receptor modulator (SARM) designed to activate the androgen receptor (AR) in muscle and bone tissue while minimizing activity in prostate and sebaceous gland tissues. This tissue selectivity is achieved through differential recruitment of coactivator proteins: in skeletal muscle, SLUP-332-bound AR recruits transcriptional coactivators (SRC-1, GRIP1) that drive myogenic gene expression, while in prostate tissue, the conformational change induced by SLUP-332 binding is insufficient to recruit the tissue-specific coactivators (such as ARA70) required for prostatic gene activation.
Peer-reviewed literature on SLUP-332 Tablets is available through the National Center for Biotechnology Information (NCBI). Explore published studies, clinical trials, and reviews:
Every batch of SLUP-332 Tablets is tested by an independent third-party laboratory. The Certificate of Analysis (COA) confirms identity, purity, and sterility of each lot.
COA documents are available upon request. Contact us with your order number to receive the COA for your specific batch.
High-Performance Liquid Chromatography (HPLC) is used to verify the purity of SLUP-332 Tablets. Our products consistently test at 99%+ purity via reverse-phase HPLC.
HPLC chromatograms are available for each batch. Request your batch report.
Mass Spectrometry (MS) confirms the molecular weight and structural identity of SLUP-332 Tablets, ensuring the compound matches its expected molecular profile.
Mass spectrometry reports are available on request. Contact support for documentation.
2nd order - tysm for the fast shipping!! got here in like 5 days. product looks good, packaging was super discreet. 10/10