


Cerebrolysin (GMP) is manufactured under strict Good Manufacturing Practice (GMP) pharmaceutical standards, ensuring absolute sterility, potency, and batch-to-batch consistency for clinical-grade research and application.
| Parameter | Value |
|---|---|
| Molecular Formula | Complex Porcine Neuropeptide Blend |
| Molecular Mass | Variable (<10,000 Da) |
| Quality Grade | GMP Clinical Grade |
| Appearance | Clear amber-tinted solution |
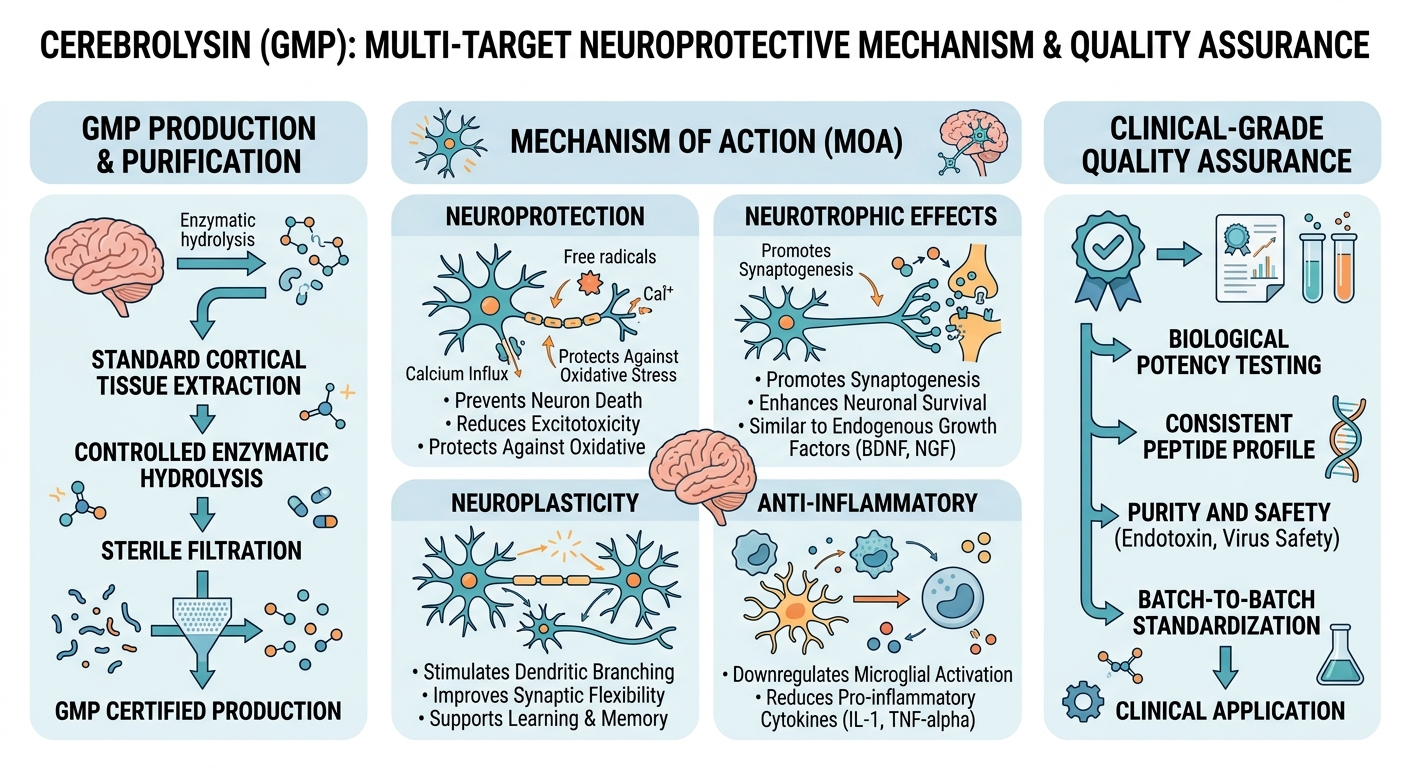
Figure 1: Cerebrolysin (GMP) Clinical Mechanism
Pharmaceutical-grade pathway delivering its primary therapeutic effect:
Figure 2: Clinical Efficacy Metrics
Cerebrolysin is a unique pharmaceutical preparation consisting of low-molecular-weight neuropeptides and free amino acids derived from enzymatic hydrolysis of purified porcine brain protein. The active fraction contains peptides below 10 kDa that mimic the activity of naturally occurring neurotrophic factors, including brain-derived neurotrophic factor (BDNF), glial cell-derived neurotrophic factor (GDNF), and ciliary neurotrophic factor (CNTF). This multi-target neurotrophic profile distinguishes it from single-compound neuroprotective agents.
Peer-reviewed literature on Cerebrolysin (GMP) is available through the National Center for Biotechnology Information (NCBI). Explore published studies, clinical trials, and reviews:
Every batch of Cerebrolysin (GMP) is tested by an independent third-party laboratory. The Certificate of Analysis (COA) confirms identity, purity, and sterility of each lot.
COA documents are available upon request. Contact us with your order number to receive the COA for your specific batch.
High-Performance Liquid Chromatography (HPLC) is used to verify the purity of Cerebrolysin (GMP). Our products consistently test at 99%+ purity via reverse-phase HPLC.
HPLC chromatograms are available for each batch. Request your batch report.
Mass Spectrometry (MS) confirms the molecular weight and structural identity of Cerebrolysin (GMP), ensuring the compound matches its expected molecular profile.
Mass spectrometry reports are available on request. Contact support for documentation.
really appreciate the batch to batch consistency! weve ordered 3 different times now and the results are basically identical each time! thats not easy to find