



BAC Water is an essential laboratory and formulation supply item required for professional peptide research and cosmetic compounding applications.
| Parameter | Value |
|---|---|
| Composition | H₂O + 0.9% Benzyl Alcohol |
| Mol. Weight | 18.02 g/mol (solvent) |
| Grade | USP Grade |
| Appearance | Crystal clear sterile solution |
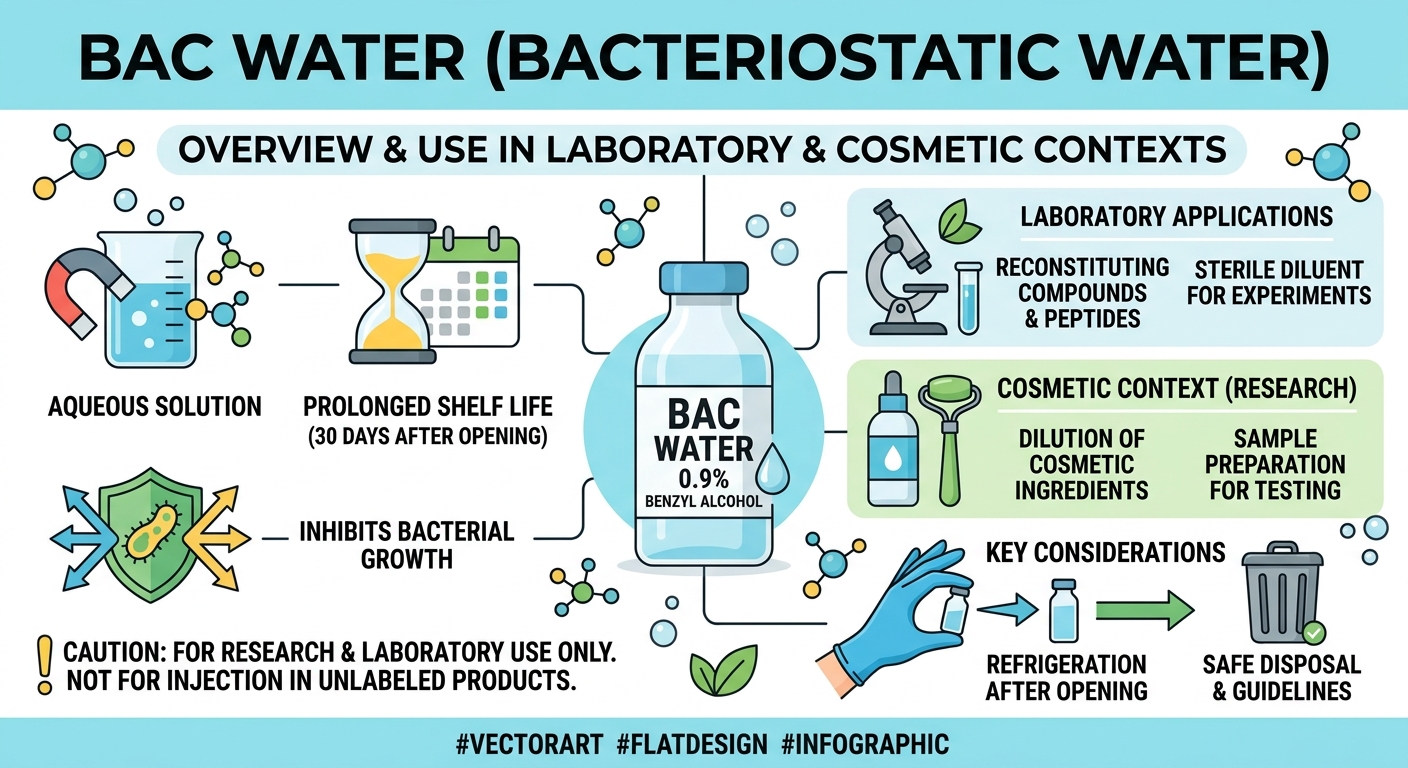
Figure 1: BAC Water Overview
Figure 2: Quality Metrics
Bacteriostatic water (BAC water) is sterile water that contains 0.9% benzyl alcohol as an antimicrobial preservative. Unlike sterile water for injection, which must be discarded after a single puncture, BAC water can be used for multiple withdrawals over a 28-day period without microbial contamination. This makes it the standard reconstitution solvent for lyophilized (freeze-dried) peptide vials in research settings.
Peer-reviewed literature on BAC Water is available through the National Center for Biotechnology Information (NCBI). Explore published studies, clinical trials, and reviews:
Every batch of BAC Water is tested by an independent third-party laboratory. The Certificate of Analysis (COA) confirms identity, purity, and sterility of each lot.
COA documents are available upon request. Contact us with your order number to receive the COA for your specific batch.
High-Performance Liquid Chromatography (HPLC) is used to verify the purity of BAC Water. Our products consistently test at 99%+ purity via reverse-phase HPLC.
HPLC chromatograms are available for each batch. Request your batch report.
Mass Spectrometry (MS) confirms the molecular weight and structural identity of BAC Water, ensuring the compound matches its expected molecular profile.
Mass spectrometry reports are available on request. Contact support for documentation.
Got here in under a week. Packaging was solid and everything was intact.